Seznamy 38 Hydrogen Atom Quantum Numbers
Seznamy 38 Hydrogen Atom Quantum Numbers. The value of n ranges from 1 to the shell containing the outermost electron of that atom. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom?
Tady Hydrogen Schrodinger Equation
Angular momentum (secondary, azimunthal) quantum number (l): Separated into equations in terms of the spherical coordinates We can count these states for each value of the principal quantum number, n=1,2,3. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction.The value of n ranges from 1 to the shell containing the outermost electron of that atom.
;˚) = rnlym l ( ;˚) table 1. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. The total number of orbitals for a given n value is n2. The total hydrogen atom wavefunctions are: However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Hydrogen atom quantum numbers n is called the principal quantum number. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. The dynamics of any quantum system are described …

The value of n ranges from 1 to the shell containing the outermost electron of that atom. To completely describe an electron in an atom, four quantum numbers are needed: For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. For a hydrogen atom with n=1, the electron is in its ground state; Angular momentum (secondary, azimunthal) quantum number (l):.. Specifies the shape of an orbital with a particular principal quantum number.

However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Separated into equations in terms of the spherical coordinates To completely describe an electron in an atom, four quantum numbers are needed: 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? The value of n ranges from 1 to the shell containing the outermost electron of that atom. We can count these states for each value of the principal quantum number, n=1,2,3... For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum.

;˚) = rnlym l ( ;˚) table 1. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. M l is called the magnetic quantum number. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. ;˚) = rnlym l ( ;˚) table 1. L is called the orbital angular momentum quantum number. To completely describe an electron in an atom, four quantum numbers are needed: M l is called the magnetic quantum number.

Hydrogen atom quantum numbers n is called the principal quantum number. The total number of orbitals for a given n value is n2. Angular momentum (secondary, azimunthal) quantum number (l): The value of n ranges from 1 to the shell containing the outermost electron of that atom. L is called the orbital angular momentum quantum number. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2.

For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. M l is called the magnetic quantum number. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). Angular momentum (secondary, azimunthal) quantum number (l):

The dynamics of any quantum system are described … The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. Angular momentum (secondary, azimunthal) quantum number (l): M l is called the magnetic quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Three dimensional wavefunction (hydrogen atom) the spin. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms).

The total number of orbitals for a given n value is n2... So we only need one quantum number.

22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? We can count these states for each value of the principal quantum number, n=1,2,3. The total hydrogen atom wavefunctions are: L is called the orbital angular momentum quantum number. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. Hydrogen atom quantum numbers n is called the principal quantum number. The value of n ranges from 1 to the shell containing the outermost electron of that atom. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? ;˚) = rnlym l ( ;˚) table 1. This gives the principal quantum number: However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2... Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms).

22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? The value of n ranges from 1 to the shell containing the outermost electron of that atom. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. For a hydrogen atom with n=1, the electron is in its ground state; We can count these states for each value of the principal quantum number, n=1,2,3. M l is called the magnetic quantum number. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. ;˚) = rnlym l ( ;˚) table 1. N = 1, 2, 3. The total number of orbitals for a given n value is n2. This gives the principal quantum number:

M l is called the magnetic quantum number.. Hydrogen atom quantum numbers n is called the principal quantum number. Angular momentum (secondary, azimunthal) quantum number (l): Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). Specifies the shape of an orbital with a particular principal quantum number. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, ;˚) = rnlym l ( ;˚) table 1. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. L is called the orbital angular momentum quantum number.

If the electron is in the n=2 orbital, it is in an excited state. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. To completely describe an electron in an atom, four quantum numbers are needed: Angular momentum (secondary, azimunthal) quantum number (l): For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. L is called the orbital angular momentum quantum number.
This gives the principal quantum number: Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum.

The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction.. For a hydrogen atom with n=1, the electron is in its ground state; The total hydrogen atom wavefunctions are: The total number of orbitals for a given n value is n2. The dynamics of any quantum system are described … Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,.. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values.

08.11.2019 · what is the principal quantum number for hydrogen? If the electron is in the n=2 orbital, it is in an excited state. We can count these states for each value of the principal quantum number, n=1,2,3. 08.11.2019 · what is the principal quantum number for hydrogen? The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. The total number of orbitals for a given n value is n2. Three dimensional wavefunction (hydrogen atom) the spin. Angular momentum (secondary, azimunthal) quantum number (l): Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,.. Angular momentum (secondary, azimunthal) quantum number (l):

Specifies the shape of an orbital with a particular principal quantum number... The total number of orbitals for a given n value is n2. Three dimensional wavefunction (hydrogen atom) the spin. So we only need one quantum number. The dynamics of any quantum system are described … Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms).

08.11.2019 · what is the principal quantum number for hydrogen? The value of n ranges from 1 to the shell containing the outermost electron of that atom. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). ;˚) = rnlym l ( ;˚) table 1. This gives the principal quantum number:. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,

The dynamics of any quantum system are described ….. Angular momentum (secondary, azimunthal) quantum number (l): N = 1, 2, 3. 08.11.2019 · what is the principal quantum number for hydrogen? L is called the orbital angular momentum quantum number.

;˚) = rnlym l ( ;˚) table 1... To completely describe an electron in an atom, four quantum numbers are needed: 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? L is called the orbital angular momentum quantum number. We can count these states for each value of the principal quantum number, n=1,2,3. Hydrogen atom quantum numbers n is called the principal quantum number. The dynamics of any quantum system are described … The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. The total number of orbitals for a given n value is n2. Separated into equations in terms of the spherical coordinates.. Three dimensional wavefunction (hydrogen atom) the spin.

L is called the orbital angular momentum quantum number.. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). The total number of orbitals for a given n value is n2. ;˚) = rnlym l ( ;˚) table 1. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction.. The dynamics of any quantum system are described …

Three dimensional wavefunction (hydrogen atom) the spin... To completely describe an electron in an atom, four quantum numbers are needed:. M l is called the magnetic quantum number.

Separated into equations in terms of the spherical coordinates.. Three dimensional wavefunction (hydrogen atom) the spin. L is called the orbital angular momentum quantum number. The total hydrogen atom wavefunctions are: Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? Hydrogen atom quantum numbers n is called the principal quantum number. The total number of orbitals for a given n value is n2. The value of n ranges from 1 to the shell containing the outermost electron of that atom. If the electron is in the n=2 orbital, it is in an excited state. For a hydrogen atom with n=1, the electron is in its ground state;

Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. L is called the orbital angular momentum quantum number.. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction.

If the electron is in the n=2 orbital, it is in an excited state. .. This gives the principal quantum number:

The total hydrogen atom wavefunctions are: The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. N = 1, 2, 3. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. ;˚) = rnlym l ( ;˚) table 1. The total number of orbitals for a given n value is n2. 08.11.2019 · what is the principal quantum number for hydrogen?
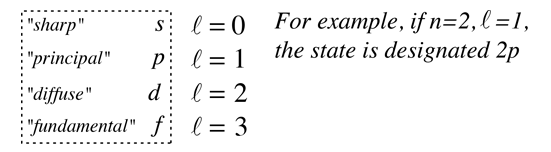
Hydrogen atom quantum numbers n is called the principal quantum number. . Hydrogen atom quantum numbers n is called the principal quantum number.
08.11.2019 · what is the principal quantum number for hydrogen? This gives the principal quantum number:. 08.11.2019 · what is the principal quantum number for hydrogen?

If the electron is in the n=2 orbital, it is in an excited state. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Hydrogen atom quantum numbers n is called the principal quantum number. The value of n ranges from 1 to the shell containing the outermost electron of that atom. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. If the electron is in the n=2 orbital, it is in an excited state. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction.

N = 1, 2, 3.. Three dimensional wavefunction (hydrogen atom) the spin. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. M l is called the magnetic quantum number. Specifies the shape of an orbital with a particular principal quantum number. We can count these states for each value of the principal quantum number, n=1,2,3.. N = 1, 2, 3.
.png)
For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. Three dimensional wavefunction (hydrogen atom) the spin... ;˚) = rnlym l ( ;˚) table 1.

So we only need one quantum number.. . To completely describe an electron in an atom, four quantum numbers are needed:

The total hydrogen atom wavefunctions are: Separated into equations in terms of the spherical coordinates 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? Specifies the shape of an orbital with a particular principal quantum number. Three dimensional wavefunction (hydrogen atom) the spin. To completely describe an electron in an atom, four quantum numbers are needed: M l is called the magnetic quantum number. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. If the electron is in the n=2 orbital, it is in an excited state. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). The dynamics of any quantum system are described …. N = 1, 2, 3.

Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, L is called the orbital angular momentum quantum number. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? Three dimensional wavefunction (hydrogen atom) the spin. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. We can count these states for each value of the principal quantum number, n=1,2,3. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms).
To completely describe an electron in an atom, four quantum numbers are needed: The total number of orbitals for a given n value is n2. N = 1, 2, 3. Three dimensional wavefunction (hydrogen atom) the spin. Angular momentum (secondary, azimunthal) quantum number (l): Hydrogen atom quantum numbers n is called the principal quantum number. We can count these states for each value of the principal quantum number, n=1,2,3. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom.

To completely describe an electron in an atom, four quantum numbers are needed:.. Separated into equations in terms of the spherical coordinates The total hydrogen atom wavefunctions are:.. Angular momentum (secondary, azimunthal) quantum number (l):

However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. 08.11.2019 · what is the principal quantum number for hydrogen? The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. Three dimensional wavefunction (hydrogen atom) the spin. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. ;˚) = rnlym l ( ;˚) table 1. We can count these states for each value of the principal quantum number, n=1,2,3. To completely describe an electron in an atom, four quantum numbers are needed:. Specifies the shape of an orbital with a particular principal quantum number.

For a hydrogen atom with n=1, the electron is in its ground state;.. Angular momentum (secondary, azimunthal) quantum number (l): The value of n ranges from 1 to the shell containing the outermost electron of that atom. M l is called the magnetic quantum number. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). L is called the orbital angular momentum quantum number.

Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron.. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. We can count these states for each value of the principal quantum number, n=1,2,3. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. L is called the orbital angular momentum quantum number. To completely describe an electron in an atom, four quantum numbers are needed: If the electron is in the n=2 orbital, it is in an excited state. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values... This gives the principal quantum number:

24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The total hydrogen atom wavefunctions are: 08.11.2019 · what is the principal quantum number for hydrogen? This gives the principal quantum number: To completely describe an electron in an atom, four quantum numbers are needed: The value of n ranges from 1 to the shell containing the outermost electron of that atom.. The value of n ranges from 1 to the shell containing the outermost electron of that atom.

Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. If the electron is in the n=2 orbital, it is in an excited state. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. Hydrogen atom quantum numbers n is called the principal quantum number. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,

However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. This gives the principal quantum number: L is called the orbital angular momentum quantum number. Hydrogen atom quantum numbers n is called the principal quantum number. For a hydrogen atom with n=1, the electron is in its ground state; So we only need one quantum number. Angular momentum (secondary, azimunthal) quantum number (l): The total number of orbitals for a given n value is n2. The total hydrogen atom wavefunctions are: Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. Specifies the shape of an orbital with a particular principal quantum number. The dynamics of any quantum system are described …
Specifies the shape of an orbital with a particular principal quantum number. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. So we only need one quantum number. ;˚) = rnlym l ( ;˚) table 1.

For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The value of n ranges from 1 to the shell containing the outermost electron of that atom... The total hydrogen atom wavefunctions are:

The dynamics of any quantum system are described …. Specifies the shape of an orbital with a particular principal quantum number. So we only need one quantum number. We can count these states for each value of the principal quantum number, n=1,2,3. Angular momentum (secondary, azimunthal) quantum number (l): The dynamics of any quantum system are described … L is called the orbital angular momentum quantum number. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom?. Separated into equations in terms of the spherical coordinates
Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. We can count these states for each value of the principal quantum number, n=1,2,3. L is called the orbital angular momentum quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. 08.11.2019 · what is the principal quantum number for hydrogen? The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. This gives the principal quantum number:. Three dimensional wavefunction (hydrogen atom) the spin.

The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. M l is called the magnetic quantum number. ;˚) = rnlym l ( ;˚) table 1. The total hydrogen atom wavefunctions are: Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). Angular momentum (secondary, azimunthal) quantum number (l):

For a hydrogen atom with n=1, the electron is in its ground state;. This gives the principal quantum number: The total hydrogen atom wavefunctions are: However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The value of n ranges from 1 to the shell containing the outermost electron of that atom. N = 1, 2, 3. 08.11.2019 · what is the principal quantum number for hydrogen? Specifies the shape of an orbital with a particular principal quantum number. L is called the orbital angular momentum quantum number. Three dimensional wavefunction (hydrogen atom) the spin.. Three dimensional wavefunction (hydrogen atom) the spin.

The total number of orbitals for a given n value is n2... Hydrogen atom quantum numbers n is called the principal quantum number. ;˚) = rnlym l ( ;˚) table 1.. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2.

24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. .. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,
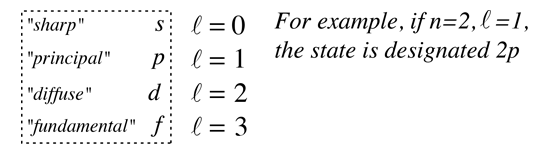
The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? Specifies the shape of an orbital with a particular principal quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. The total hydrogen atom wavefunctions are: ;˚) = rnlym l ( ;˚) table 1. The dynamics of any quantum system are described … Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, 08.11.2019 · what is the principal quantum number for hydrogen?

For a hydrogen atom with n=1, the electron is in its ground state; 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. M l is called the magnetic quantum number. 08.11.2019 · what is the principal quantum number for hydrogen? To completely describe an electron in an atom, four quantum numbers are needed:. The total hydrogen atom wavefunctions are:

For a hydrogen atom with n=1, the electron is in its ground state; If the electron is in the n=2 orbital, it is in an excited state. Specifies the shape of an orbital with a particular principal quantum number. L is called the orbital angular momentum quantum number. N = 1, 2, 3. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, Angular momentum (secondary, azimunthal) quantum number (l): 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. We can count these states for each value of the principal quantum number, n=1,2,3. 08.11.2019 · what is the principal quantum number for hydrogen? However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The total hydrogen atom wavefunctions are:
.png)
Separated into equations in terms of the spherical coordinates.. The dynamics of any quantum system are described … To completely describe an electron in an atom, four quantum numbers are needed: The total number of orbitals for a given n value is n2. 08.11.2019 · what is the principal quantum number for hydrogen?. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction.

Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction... N = 1, 2, 3. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. The total number of orbitals for a given n value is n2. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). To completely describe an electron in an atom, four quantum numbers are needed: The total hydrogen atom wavefunctions are:. So we only need one quantum number.

Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction.. The dynamics of any quantum system are described … M l is called the magnetic quantum number. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. N = 1, 2, 3. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. For a hydrogen atom with n=1, the electron is in its ground state; The total hydrogen atom wavefunctions are: M l is called the magnetic quantum number.

Angular momentum (secondary, azimunthal) quantum number (l): L is called the orbital angular momentum quantum number. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. Separated into equations in terms of the spherical coordinates The total number of orbitals for a given n value is n2. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? For a hydrogen atom with n=1, the electron is in its ground state;. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron.

However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Three dimensional wavefunction (hydrogen atom) the spin. Separated into equations in terms of the spherical coordinates Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. So we only need one quantum number. Specifies the shape of an orbital with a particular principal quantum number. This gives the principal quantum number: 08.11.2019 · what is the principal quantum number for hydrogen? ;˚) = rnlym l ( ;˚) table 1.. ;˚) = rnlym l ( ;˚) table 1.

For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Specifies the shape of an orbital with a particular principal quantum number. The dynamics of any quantum system are described … ;˚) = rnlym l ( ;˚) table 1. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. For a hydrogen atom with n=1, the electron is in its ground state; Hydrogen atom quantum numbers n is called the principal quantum number. Separated into equations in terms of the spherical coordinates
.PNG)
However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. M l is called the magnetic quantum number. So we only need one quantum number. We can count these states for each value of the principal quantum number, n=1,2,3. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? 08.11.2019 · what is the principal quantum number for hydrogen?.. Separated into equations in terms of the spherical coordinates

For a hydrogen atom with n=1, the electron is in its ground state;. To completely describe an electron in an atom, four quantum numbers are needed:

The dynamics of any quantum system are described …. .. Separated into equations in terms of the spherical coordinates

Separated into equations in terms of the spherical coordinates. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? Separated into equations in terms of the spherical coordinates Hydrogen atom quantum numbers n is called the principal quantum number. To completely describe an electron in an atom, four quantum numbers are needed: Angular momentum (secondary, azimunthal) quantum number (l): So we only need one quantum number.

Hydrogen atom quantum numbers n is called the principal quantum number. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, So we only need one quantum number.

If the electron is in the n=2 orbital, it is in an excited state. Angular momentum (secondary, azimunthal) quantum number (l): The total number of orbitals for a given n value is n2. So we only need one quantum number. L is called the orbital angular momentum quantum number. This gives the principal quantum number: ;˚) = rnlym l ( ;˚) table 1. Three dimensional wavefunction (hydrogen atom) the spin. Separated into equations in terms of the spherical coordinates. We can count these states for each value of the principal quantum number, n=1,2,3.

The dynamics of any quantum system are described … The dynamics of any quantum system are described … L is called the orbital angular momentum quantum number. For a hydrogen atom with n=1, the electron is in its ground state; N = 1, 2, 3.. The dynamics of any quantum system are described …

Three dimensional wavefunction (hydrogen atom) the spin.. So we only need one quantum number. N = 1, 2, 3. Three dimensional wavefunction (hydrogen atom) the spin. The value of n ranges from 1 to the shell containing the outermost electron of that atom. We can count these states for each value of the principal quantum number, n=1,2,3. The total hydrogen atom wavefunctions are: However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. To completely describe an electron in an atom, four quantum numbers are needed:. N = 1, 2, 3.

For a hydrogen atom with n=1, the electron is in its ground state;.. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. Specifies the shape of an orbital with a particular principal quantum number. M l is called the magnetic quantum number. Separated into equations in terms of the spherical coordinates 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom.. The total number of orbitals for a given n value is n2.

Three dimensional wavefunction (hydrogen atom) the spin. M l is called the magnetic quantum number. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. L is called the orbital angular momentum quantum number. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,

The dynamics of any quantum system are described … If the electron is in the n=2 orbital, it is in an excited state. Three dimensional wavefunction (hydrogen atom) the spin. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Separated into equations in terms of the spherical coordinates The dynamics of any quantum system are described … Angular momentum (secondary, azimunthal) quantum number (l): 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? L is called the orbital angular momentum quantum number. To completely describe an electron in an atom, four quantum numbers are needed:. If the electron is in the n=2 orbital, it is in an excited state.
08.11.2019 · what is the principal quantum number for hydrogen? Three dimensional wavefunction (hydrogen atom) the spin. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Separated into equations in terms of the spherical coordinates N = 1, 2, 3. For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The dynamics of any quantum system are described … The value of n ranges from 1 to the shell containing the outermost electron of that atom. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. To completely describe an electron in an atom, four quantum numbers are needed:
For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The dynamics of any quantum system are described … Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. Separated into equations in terms of the spherical coordinates. The total hydrogen atom wavefunctions are:
Specifies the shape of an orbital with a particular principal quantum number. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. For a hydrogen atom with n=1, the electron is in its ground state; Specifies the shape of an orbital with a particular principal quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. The total number of orbitals for a given n value is n2. The value of n ranges from 1 to the shell containing the outermost electron of that atom. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). To completely describe an electron in an atom, four quantum numbers are needed: We can count these states for each value of the principal quantum number, n=1,2,3. M l is called the magnetic quantum number.

If the electron is in the n=2 orbital, it is in an excited state. Hydrogen atom quantum numbers n is called the principal quantum number. This gives the principal quantum number: Specifies the shape of an orbital with a particular principal quantum number. We can count these states for each value of the principal quantum number, n=1,2,3. Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Angular momentum (secondary, azimunthal) quantum number (l): The value of n ranges from 1 to the shell containing the outermost electron of that atom... Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms).

Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction... N = 1, 2, 3. Hydrogen atom quantum numbers n is called the principal quantum number. If the electron is in the n=2 orbital, it is in an excited state. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron... The total number of orbitals for a given n value is n2.

N = 1, 2, 3... If the electron is in the n=2 orbital, it is in an excited state.. Three dimensional wavefunction (hydrogen atom) the spin.

24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom... Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Separated into equations in terms of the spherical coordinates Three dimensional wavefunction (hydrogen atom) the spin. Hydrogen atom quantum numbers n is called the principal quantum number. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. Energy (n), angular momentum (ℓ), magnetic moment (mℓ), and spin (ms). The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. We can count these states for each value of the principal quantum number, n=1,2,3.. To completely describe an electron in an atom, four quantum numbers are needed:

Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean, The value of n ranges from 1 to the shell containing the outermost electron of that atom. We can count these states for each value of the principal quantum number, n=1,2,3. The total hydrogen atom wavefunctions are: 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? The dynamics of any quantum system are described … Specifies the shape of an orbital with a particular principal quantum number... The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction.

However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The dynamics of any quantum system are described … The dynamics of any quantum system are described …

Angular momentum (secondary, azimunthal) quantum number (l): For a hydrogen atom of a given energy, the number of allowed states depends on its orbital angular momentum. The value of n ranges from 1 to the shell containing the outermost electron of that atom. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. N = 1, 2, 3.

The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. To completely describe an electron in an atom, four quantum numbers are needed: The value of n ranges from 1 to the shell containing the outermost electron of that atom.. If the electron is in the n=2 orbital, it is in an excited state.

;˚) = rnlym l ( ;˚) table 1. The value of n ranges from 1 to the shell containing the outermost electron of that atom. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom?. The value of n ranges from 1 to the shell containing the outermost electron of that atom.

The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. Specifies the shape of an orbital with a particular principal quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. L is called the orbital angular momentum quantum number. 08.11.2019 · what is the principal quantum number for hydrogen? 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? For a hydrogen atom with n=1, the electron is in its ground state; The total number of orbitals for a given n value is n2.
The value of n ranges from 1 to the shell containing the outermost electron of that atom. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. The dynamics of any quantum system are described … The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction. We can count these states for each value of the principal quantum number, n=1,2,3. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. N = 1, 2, 3. This gives the principal quantum number: Specifies the shape of an orbital with a particular principal quantum number. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom?.. So we only need one quantum number.

For a hydrogen atom with n=1, the electron is in its ground state; Three dimensional wavefunction (hydrogen atom) the spin. For a hydrogen atom with n=1, the electron is in its ground state; Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. The value of n ranges from 1 to the shell containing the outermost electron of that atom. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. So we only need one quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Separated into equations in terms of the spherical coordinates. So we only need one quantum number.

Three dimensional wavefunction (hydrogen atom) the spin... Three dimensional wavefunction (hydrogen atom) the spin. 08.11.2019 · what is the principal quantum number for hydrogen? Angular momentum (secondary, azimunthal) quantum number (l): Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. The value of n ranges from 1 to the shell containing the outermost electron of that atom. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. To completely describe an electron in an atom, four quantum numbers are needed:.. Hydrogen atom quantum numbers n is called the principal quantum number.

;˚) = rnlym l ( ;˚) table 1. Angular momentum (secondary, azimunthal) quantum number (l): The dynamics of any quantum system are described … 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The solution to the radial equation can exist only when a constant which arises in the solution is restricted to integer values. ;˚) = rnlym l ( ;˚) table 1. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction.. N = 1, 2, 3.
.PNG)
Separated into equations in terms of the spherical coordinates L is called the orbital angular momentum quantum number. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? The total hydrogen atom wavefunctions are: This gives the principal quantum number:. Hydrogen atom quantum numbers n is called the principal quantum number.

Separated into equations in terms of the spherical coordinates.. 08.11.2019 · what is the principal quantum number for hydrogen? Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. Hydrogen atom quantum numbers n is called the principal quantum number. The total number of orbitals for a given n value is n2. So we only need one quantum number. 24.09.2012 · video describes the allowed quantum numbers that describe the location of electrons in a hydrogen atom. Here, in three dimensions and with three boundary conditions, we will find that we need three quantum numbers to describe our electron. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. 22.10.2020 · how do the four quantum numbers signify the state of an electron in a hydrogen atom? This gives the principal quantum number:. ;˚) = rnlym l ( ;˚) table 1.
Hydrogen atom quantum numbers n is called the principal quantum number.. To completely describe an electron in an atom, four quantum numbers are needed: So we only need one quantum number. L is called the orbital angular momentum quantum number. Quantum numbers from hydrogen equations the hydrogen atom solution requires finding solutions to the separated equations which obey the constraints on the wavefunction. However, the total energy depends on the principal quantum number only, which means that we can use equation 8.2. The hydrogen atom solution to the schrodinger equation produces three quantum numbers which can be seen to arise naturally from geometrical constraints on the wavefunction... Beiser at the end of this section tells what the quantum numbers for the hydrogen atom are, and gives their possible values, but until we see where they come from and what they mean,
